Purdue Pharma left almost nothing to chance in the whirlwind marketing of its new painkiller OxyContin.
From 1996 to 2002, Purdue pursued nearly every avenue in the drug supply and prescription sales chain—a strategy now cast as reckless and illegal in more than 1,500 federal civil lawsuits from communities in Florida to Wisconsin to California that allege the drug has fueled a national epidemic of addiction.
Kaiser Health News is releasing years of Purdue’s internal budget documents and other records to offer readers a chance to evaluate how the privately held Connecticut company spent hundreds of millions of dollars to launch and promote the drug, a trove of information made publicly available here for the first time.
All of these internal Purdue records were obtained from a Florida attorney general’s office investigation of Purdue’s sales efforts that ended late in 2002.
I have had copies of those records in my basement for years. I was a reporter at the South Florida Sun-Sentinel, which, along with the Orlando Sentinel, won a court battle to force the attorney general to release the company files in 2003. At the time, the Sun-Sentinel was writing extensively about a growing tide of deaths from prescription drugs such as OxyContin.
We drew on the marketing files to write two articles, including one that exposed possible deceptive marketing of the drug. Now, given the disastrous arc of prescription drug abuse over the past decade and the stream of suits being filed — more than a dozen on some days — it seemed time for me to share these seminal documents that reveal the breadth and detail of Purdue’s efforts.
Asked by Kaiser Health News for comment on the OxyContin marketing files and the suits against the company, Purdue Pharma spokesman Robert Josephson issued a statement that reads in part:
“Suggesting activities that last occurred more than 16 years ago, for which the company accepted responsibility, helped contribute to today’s complex and multi-faceted opioid crisis is deeply flawed. The bulk of opioid prescriptions are not, and have never been, for OxyContin, which represents less than 2% of current opioid prescriptions.”
The backdrop
The marketing files show that about 75 percent of more than $400 million in promotional spending occurred after the start of 2000, the year Purdue officials told Congress they learned of growing OxyContin abuse and drug-related deaths from media reports and regulators.
These internal Purdue marketing records show the drugmaker financed activities across nearly every quarter of medicine, from awarding grants to health care groups that set standards for opioid use to reminding reluctant pharmacists how they could profit from stocking OxyContin pills on their shelves.
Purdue bought more than $18 million worth of advertising in major medical journals that cheerily touted OxyContin. Some of the ads, federal officials said in 2003, “grossly overstated” the drug’s safety.
The Purdue records show that the company poured more than $8 million into a website and venture called “Partners Against Pain,” which helped connect patients to doctors willing to treat their pain, presumably with OxyContin or other opioids.
It made and distributed 14,000 copies of a video that claimed opioids caused addiction in fewer than 1 percent of patients, a claim Food and Drug Administration officials later said “has not been substantiated.”
Purdue hoped to grow into one of the nation’s top 10 drug companies, both in sales and “image or professional standing,” according to the documents; OxyContin was the means to that end.
Purdue first marketed the drug for cancer pain but planned to expand that use to meet its multimillion-dollar sales goals. In 1998, the market for treating cancer with opioids stood at $261 million, compared with $1.3 billion for treating other types of pain, the Purdue reports note.
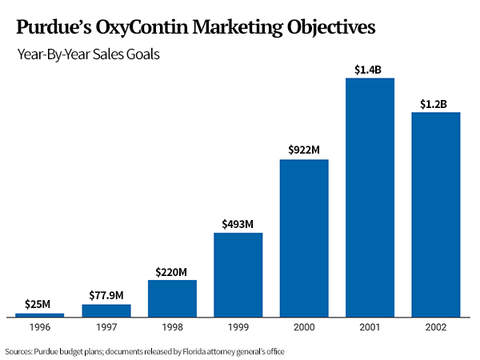
Purdue’s OxyContin sales objectives were clearly stated in the earliest marketing plan in the records, for 1996. It sought $25 million in sales and to generate 205,000 prescriptions. By the next year, its goals had tripled: $77.9 million in sales and to generate 600,000 prescriptions.
The strategy
Purdue bombarded doctors and other health workers with literature and sales calls. Records show that in 1997 the company budgeted $300,000 for mailings to doctors who prescribed opioids liberally, based on sales data that drug companies purchase. The mailers recommended OxyContin for “pain syndromes,” including osteoarthritis and back pain. It added $75,000 for mailings “to keep in touch with our best customers for OxyContin to ensure they continue prescribing it.”
Sales agents made thousands of visits to general practice doctors and others who had little training or experience using potent opioids, according to a 2003 Government Accountability Office audit. The OxyContin slogan in 1999 was: “The One to Start With and the One to Stay With.” OxyContin earned Purdue about $2.8 billion in revenue from the start of 1996 through June 2001, according to the Justice Department.
In May 2000, Purdue’s hope to conquer the arthritis market hit a snag when the FDA criticized an ad for OxyContin in the New England Journal of Medicine. The FDA said the ad, which Purdue Pharma agreed to stop using, overstated the drug’s benefits for treating all types of arthritis without pointing out risks.
News reports of abuse and overdose deaths also were surfacing. Purdue’s 2001 marketing document noted that OxyContin had “experienced significant challenges” the year before because of abuse and unlawful diversion in Maine, Ohio, Virginia, Louisiana and Florida.
OxyContin pills contain oxycodone, an opioid as potent as morphine and maybe more so. Abusers quickly figured out they could crush the pills and snort or inject the dust.
In response, Purdue’s 2001 marketing budget included funding to help doctors recognize patients who were in need of “substance abuse counseling” and do more to “prevent abuse and diversion.” It added $1.2 million in spending for what it called “anti-diversion” efforts in 2002, according to the internal records.
Potent sales force

In 2002, the Florida attorney general’s office was one of the first law enforcement agencies to investigate Purdue. The state ended its probe after Purdue agreed to pay Florida $2 million to help fund a data system to monitor narcotics prescriptions. It did not admit to any wrongdoing in the settlement.
Yet handwritten notes of a state investigator’s interview with a former Purdue sales manager for West Virginia and western Pennsylvania named Bill Gergely, then 58, suggested otherwise. The notes were part of the documents released by the state.
Gergely, who worked for the company from 1972 until 2000, said Purdue executives told sales staff at a launch meeting that OxyContin “was non-habit forming,” according to the undated investigator’s notes. Gergely said Purdue gave its sales force material — some of which was not approved by the FDA — for “education,” the notes show. He told the investigator that Purdue had a bonus system and paid well; the last year he worked for Purdue, Gergely earned $238,000.
As Purdue charged ahead with OxyContin, prescription pills overtook illegal drugs like heroin and cocaine as killers in Florida, according to medical examiner files. In May 2002, the South Florida Sun-Sentinel documented nearly 400 pill deaths in three South Florida counties the previous two years, based on an examination of autopsy and police records.
Half the deaths involved drugs that contained oxycodone, according to medical examiner records. But it was not always clear in these records that it was OxyContin because oxycodone was an ingredient in many other narcotic pills. In 70 of the deaths, however, police or medical examiner records specifically identified OxyContin as one of the drugs. Though some people who died bought pills on a thriving black market, many were under the care of doctors for what appeared, at least at some point, to be legitimate injuries, according to medical examiner files.
Purdue did not challenge the accuracy of the newspaper’s reporting. It countered that the articles “did a disservice” to the company and patients who take their medicine “according to the directions of their doctors.” While the company said its executives “deeply regret the tragic consequences that have resulted from the misuse and abuse of our pain medicine … advances in the treatment of pain should not be limited or reversed because some people illegally divert, abuse or misuse these drugs.”
To its sales force, the internal Purdue records show, Purdue blamed bad press for cutting into sales. “The media’s attention to abuse and diversion of OxyContin tablets has provided state Medicaid plans and some HMOs, concerned about the effect the product is having on their budget, an excuse to look for ways to limit the prescribing of OxyContin tablets,” the 2002 marketing document said.
But five years after its legal battle with Florida officials, Purdue made a startling admission in federal court in Virginia. The company pleaded guilty in 2007 to felony charges of “misbranding” OxyContin “with the intent to defraud or mislead.” The company paid $600 million in fines and other penalties. Among the deceptions it confessed to was directing its salespeople to tell doctors the drug was less addictive than other opioids.
Three Purdue Pharma executives pleaded guilty to misdemeanor criminal charges for their roles in the marketing scheme. The three men paid a total of $34 million in fines and penalties, court records show. Accepting Purdue’s plea deal, U.S. District Judge James P. Jones noted that federal prosecutors believed the Purdue case of 2007 would send a “strong deterrent message to the pharmaceutical industry.”
A Costly Reckoning?
Ten years on, the 1,500-plus lawsuits, filed mostly on behalf of cities, counties and states, could prove to be a costly reckoning for the opioid industry. The suits are demanding payback from Purdue and other drugmakers for the sky-high costs of treating addiction and other compensation, much as the litigation against Big Tobacco in the late 1990s.
Other drug makers named as defendants in most of the suits include those that Purdue considered to be its top competitors in the pain sector: Janssen Pharmaceuticals, Teva Pharmaceutical Industries, Endo International PLC and Mallinckrodt PLC.
Federal officials estimate the economic cost of opioid abuse topped $500 billion in 2015 alone. Since 1999, at least 200,000 people have died in the U.S. from these overdoses, according to the Centers for Disease Control and Prevention. More than 52,000 of those died in 2015 alone, more than were killed in car crashes and gun homicides combined, the suits contend.
A case filed in April by Baltimore County in Maryland makes an argument common to many of the suits:
“From the mid-’90s to the present, manufacturing defendants aggressively marketed and falsely promoted liberal opioid prescribing as presenting little to no risk of addiction, even when used long term for chronic pain. They infiltrated academic medicine and regulatory agencies to convince doctors that treating chronic pain with long-term opioids was evidence-based medicine when, in fact, it was not.
“Huge profits resulted from these efforts — as did the present addiction and overdose crisis.”
Purdue has not yet filed a response to the allegations in the suit.
Other drug manufacturers “emulated Purdue’s false marketing strategy” and sold billions of dollars of prescription opioids “as safe and efficacious for long term use, knowing full well that they were not,” Wisconsin’s Oneida County alleges in its November 2017 federal court suit. Purdue also has not yet filed a response to the allegations in this suit.
But Purdue spokesman Josephson told KHN: “We share public officials’ concern about the opioid crisis, and we are committed to working collaboratively toward meaningful solutions. We vigorously deny these allegations and look forward to the opportunity to present our defense.”
One California doctor who was sentenced to 25 years in prison for overprescribing OxyContin is also suing Purdue. Masoud Bamdad alleges that the company’s representatives made sales calls and gave him “deceitful, misleading and over-hyped information,” which he relied on to prescribe the drug, in some cases with deadly consequences for his patients, according to the suit, which is pending. Purdue has asked that the case be stayed while judges decide if it should be consolidated with others filed against the company. In February, Purdue announced that it would no longer promote opioids to doctors.
Because the lawsuits from across the U.S. contain similar allegations, many of them have been consolidated in Ohio – as a multi-district litigation. Some days, federal court dockets log a dozen or more new cases. Many of the suits run a hundred pages or more and allege that deceptive opioid marketing schemes continue to this day.
The manufacturers, in a joint court motion late last year, contend that opioids “serve a critical public health role in providing relief to patients suffering from pain that is often debilitating” and that they are being wrongly blamed.
They also point out that the FDA approved all of their products as “safe and effective.”
This month, the manufacturers filed motions to dismiss several of the cases, arguing that the county governments lack a legal basis for their claims. In seeking to blame the drugmakers, these lawsuits ignore “the criminal acts of third parties, the crucial role of health care providers, and the thorny public policy questions surrounding the problem of opioid abuse,” reads a motion to dismiss a case filed by Monroe County, Mich., against Purdue Pharma and other drug companies.
Dan Polster, the federal judge handling the cases, told an overflow crowd in his courtroom that the opioid epidemic has become so severe, that it is cutting the average life expectancy of Americans.
“I’m pretty ashamed that this has occurred while I have been around,” he said in January, adding, “I think we all should be.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation, which is not affiliated with Kaiser Permanente. KHN’s coverage of prescription drug development, costs and pricing is supported in part by the Laura and John Arnold Foundation.